Why this matters: Carbon is the backbone of life (DNA) and the future of technology (Nanotubes).
Attention Activity: The Carbon Paradox

If they are both made of the exact same atoms (Carbon), how can they be so different?
Click "Next" to unlock the secret hidden in Carbon's bonding structure.
Why Carbon Rules the World

Carbon isn't just a chemical element; it's the "LEGO brick" of the universe. Because of its unique structure, it can form millions of different compounds.
- Biological: DNA, proteins, and carbohydrates.
- Industrial: Steel, plastics, and fuels.
- Futuristic: Graphene and carbon fiber.
The Magic Number: 4
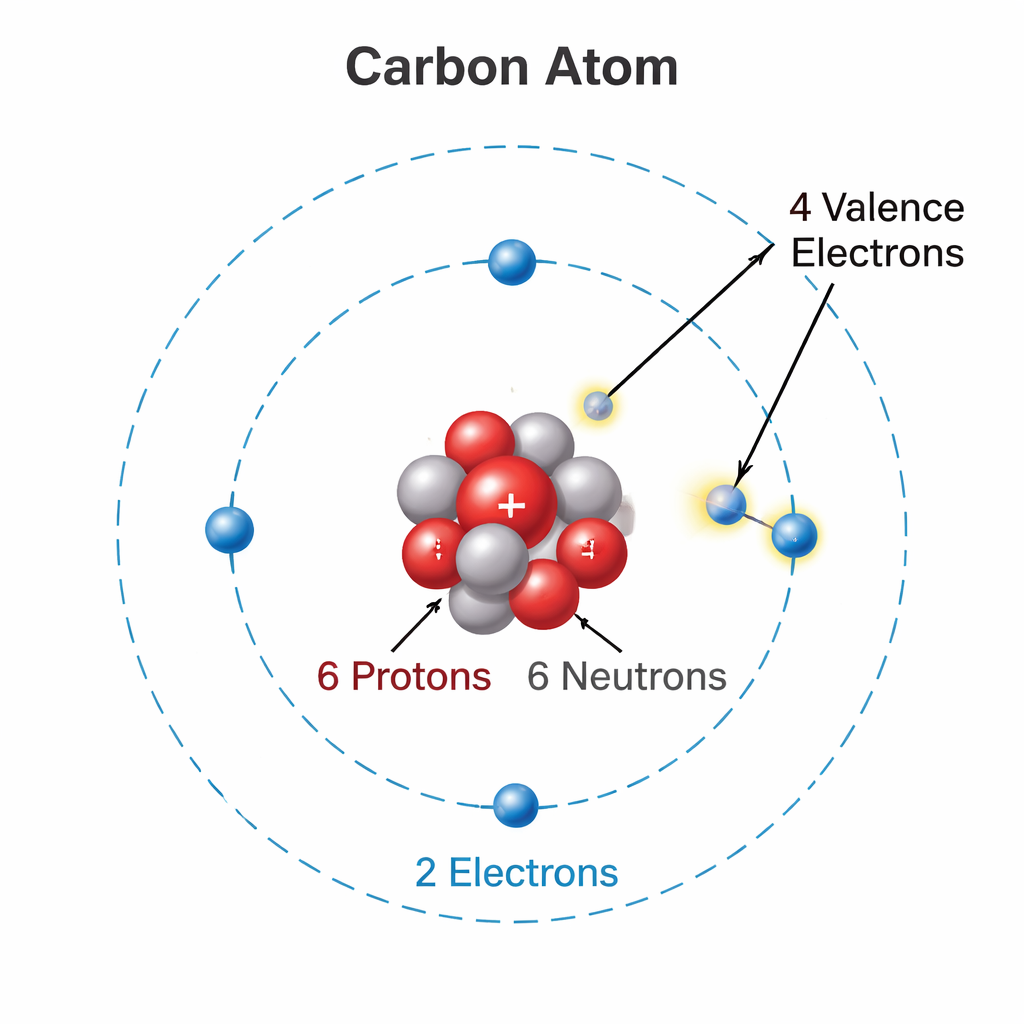
Carbon has an atomic number of 6. Its electronic configuration is (2, 4).
The solution? Carbon shares its 4 valence electrons with other atoms. This is called Covalent Bonding.
Knowledge Check 1
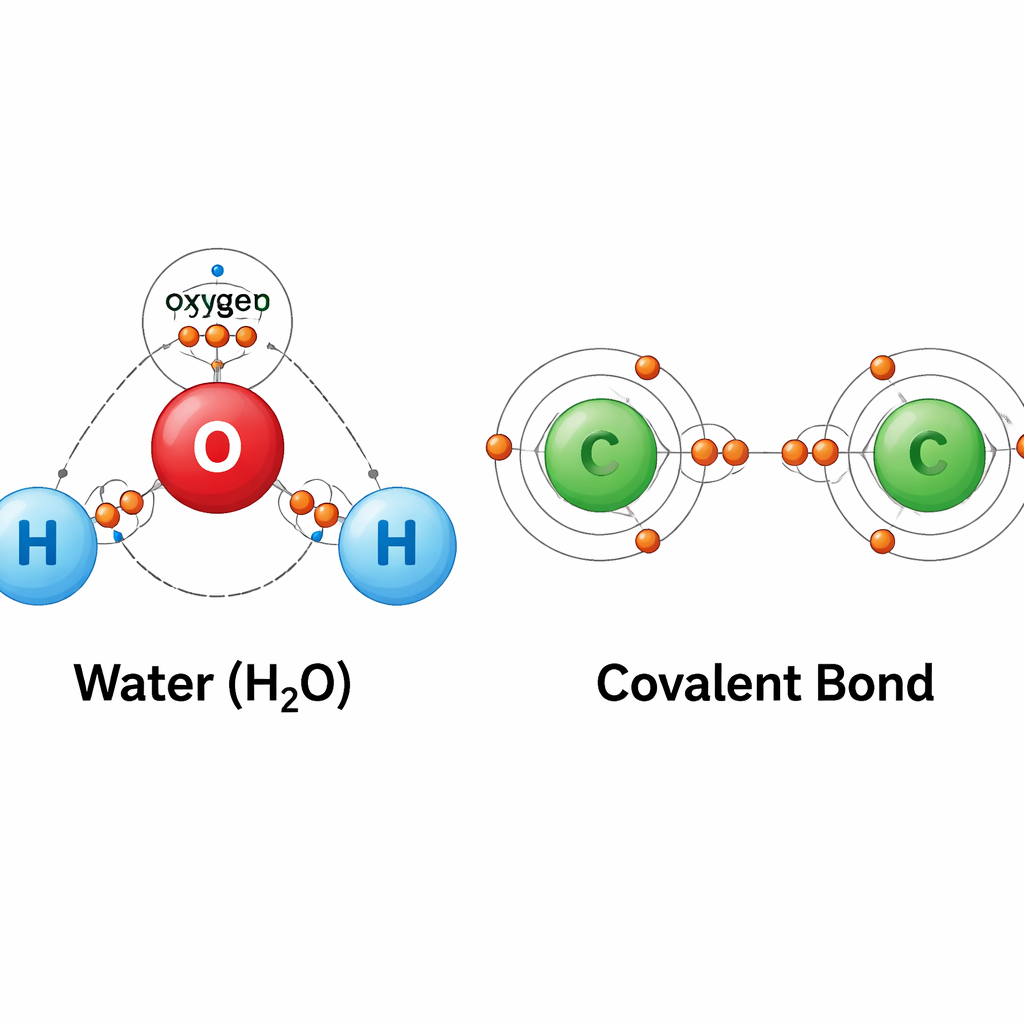
Based on the atomic structure we just discussed, why does Carbon primarily form covalent bonds?
Catenation & Tetravalency

Carbon has two "Superpowers":
- Catenation: The ability to bond with itself to form long chains or rings.
- Tetravalency: Having 4 valence electrons allows it to bond with up to 4 other atoms.
Allotropes: Diamond

In Diamond, each carbon atom is bonded to four other carbon atoms in a rigid 3D tetrahedral structure. This makes it the hardest natural substance.
Allotropes: Graphite
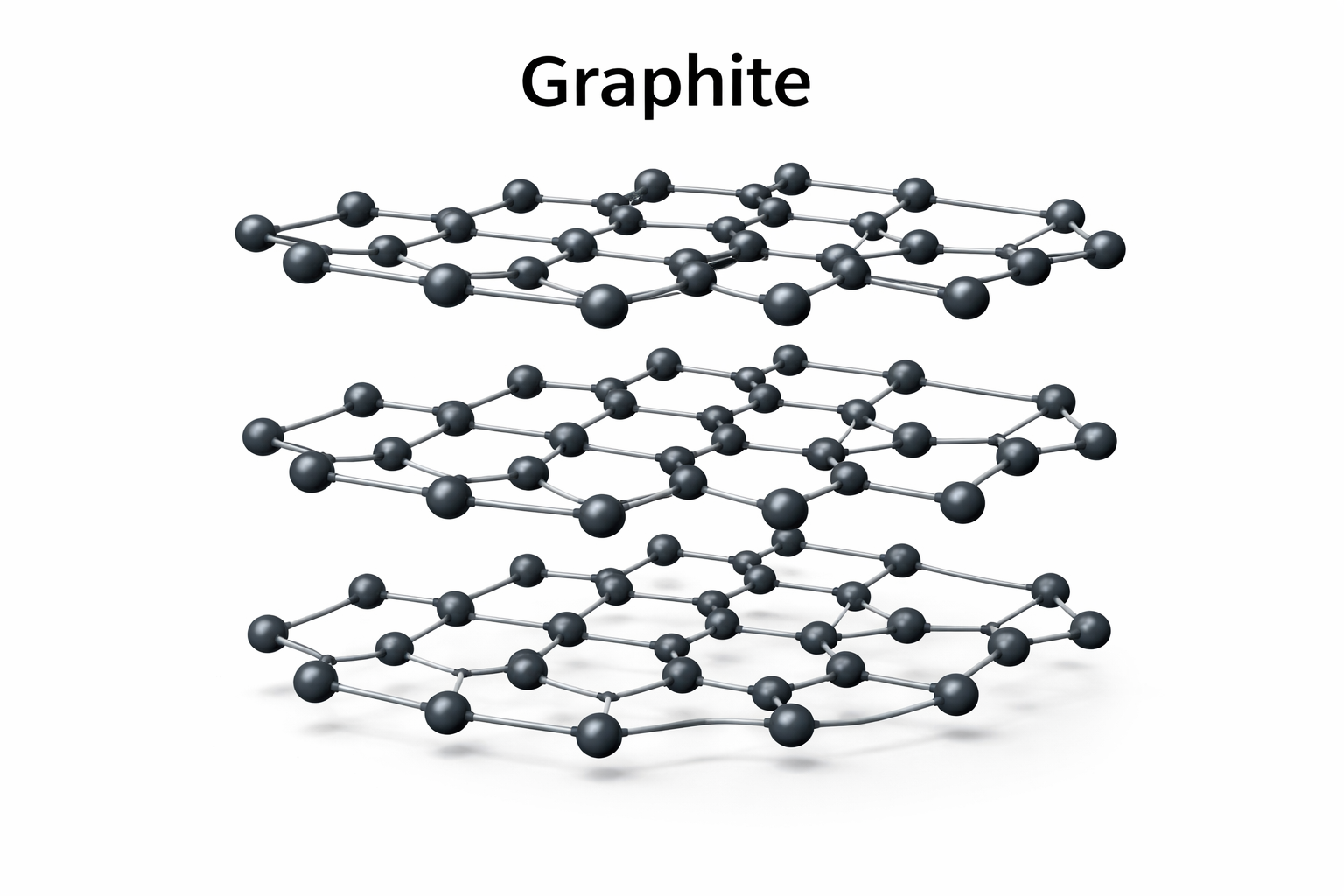
In Graphite, each atom is bonded to three others in hexagonal layers. These layers can slide over each other, making graphite soft and slippery.
Knowledge Check 2
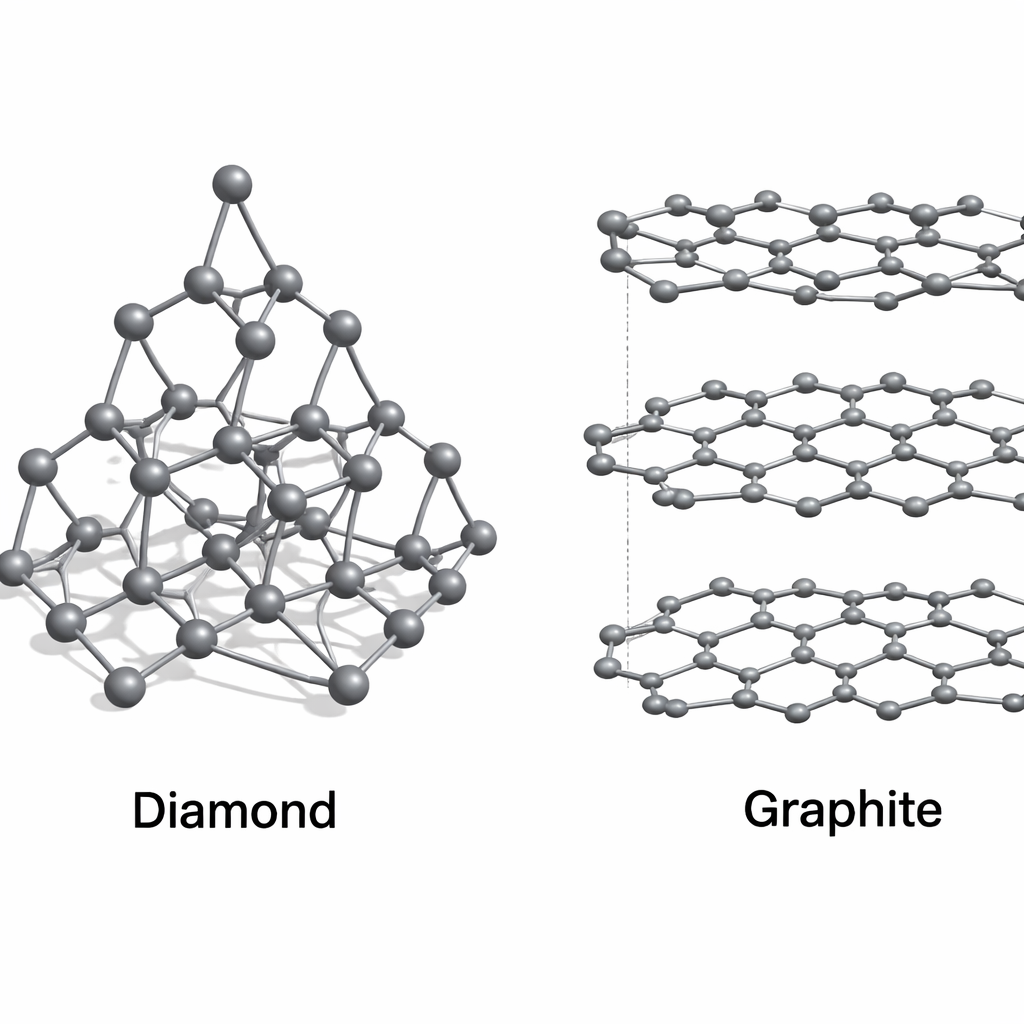
Look at the structures above. Why is Graphite a good conductor of electricity while Diamond is not?
Protocol Complete: Assessment
You've completed the tutorial. Let's verify your knowledge.
Passing Score: 80%
Key Takeaways
- Review the core ideas.
- Connect concepts to practice.
- Prepare for assessment.
Question 1
What is the electronic configuration of Carbon?
Question 2
The property of carbon to link with other carbon atoms to form long chains is called:
Question 3
In Diamond, how many bonds does each carbon atom form with other carbon atoms?
Question 4
Which carbon allotrope is used as a lubricant due to its layered structure?
Question 5
Which of the following describes C-60 Buckminsterfullerene?
Final Score
Protocol Complete: Carbon and its Compounds
This certifies that
Student
has mastered the core principles of Carbon Science.
Score: 0%
